Abstract

Background: Mutations in the epigenetic regulators or chromatin modifiers DNMT3A, TET2, and ASXL1, collectively termed DTA mutations, are common early hits in myeloid neoplasms, including myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML). Our group has recently shown the impact of pre-transplant mutation status on survival for patients with DTA-mutant clones, in addition to IDH1/2- and EZH2-mutant clones (Selove et al., eJHaem. 2022; 2:514-19). In principle, hypomethylating agent (HMA)-based therapy might specifically eliminate TET2-mutant clones. Wild-type TET2 is a demethylase, and TET2 mutation imparts a hypermethylated phenotype that could potentially be reversed by HMAs. However, this is rarely observed in the clinical setting.
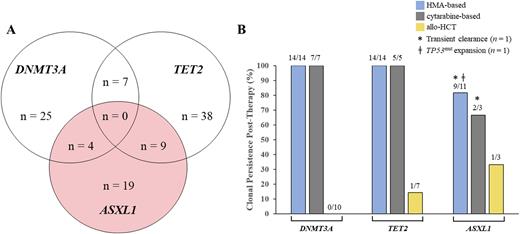
Methods: Identification of patients with DTA mutations from the UMass Chan Leukemia Registry was performed via UMass Clarity, a Microsoft SQL Server database comprised of 23,000 tables, in conjunction with the main reporting environment in EPIC. The Registry identified patients harboring one or more aberrations in the DTA cluster and with ICD-10 codes of either D46.9 (MDS, including its subentities) or C92.00 (AML, including its subentities) between 2011-2021. We evaluated the frequency of DTA mutations in 495 patients with confirmed diagnoses of myeloid malignancies, of whom 36 (7.3%) harbored DNMT3A mutation(s), 54 (10.9%) harbored TET2 mutation(s), and 32 (6.5%) harbored ASXL1 mutation(s) (Panel A). Responses to treatment were assessed through serial bone marrow examinations, which were available for 21 patients with DNMT3A mutations, 22 patients with TET2 mutations, and 14 patients with ASXL1 mutations. We investigated the effects of HMA- and cytarabine-based therapies on DTA-mutant clones to discern the effectiveness of first-line treatment.
Results:DNMT3A mutations persisted in 21 (100%) of 21 patients assessed who received first-line HMA-based therapy (n = 14) or cytarabine-based therapy (n = 7) (Panel B). In 20 (95.2%) of the 21 patients assessed with DNMT3A mutations, HMA-based (13 of 14 patients) and cytarabine-based (7 of 7 patients) therapy resulted in relative clonal enrichment of DNMT3A mutations, despite eliminating clones harboring mutations in other biological clusters. Of note, targeted therapy often extinguished the susceptible mutation but did not reduce the burden of the DNMT3A-mutant clone. In contrast, the DNMT3A-mutant clone was eradicated in all 10 (100%) of the patients who underwent allogeneic hematopoietic cell transplant (allo-HCT).
TET2 mutations persisted in 19 (100%) of 19 patients who received first-line HMA-based (n = 14) or cytarabine-based (n = 5) therapy (Panel B). The relative TET2 mutational burden increased in 16 (84.2%) of the 19 patients treated with HMA-based or cytarabine-based therapy. In 2 patients, the original TET2 mutation disappeared, but a new TET2 mutation emerged. The TET2 mutation(s) were eradicated in 6 (85.7%) of 7 patients who underwent allo-HCT.
ASXL1 mutations persisted in 9 (81.8%) of 11 patients treated with HMA and 2 (66.6%) of 3 patients treated with cytarabine-based chemotherapy (Panel B). Of the 3 patients who had clearance of the ASXL1-mutant clone, clearance was transient in 2 patients, and 1 patient had expansion of a TP53-mutant clone. In 12 (85.7%) of 14 patients treated with HMA-based therapy or cytarabine-based therapy, the relative ASXL1-mutant clonal size increased. ASXL1 mutations were eradicated in 2 (66.7%) of 3 patients who underwent allo-HCT. For ASXL1 with TP53 co-mutation, the TP53-mutant clone expanded on HMA-based therapy.
Conclusions: Neither HMA-based nor cytarabine-based therapy reliably eradicated DTA-mutant clones. In fact, the relative clonal size of DTA mutations increased after such therapy, despite morphologic remission in many cases, highlighting the resistance of DTA-mutant cells. However, subsequent allo-HCT was effective at eliminating DTA-mutant clones (including 100% of DNMT3A-mutant clones). A successful therapeutic strategy for patients with DTA mutations may therefore be initial treatment with either HMA- or cytarabine-based therapy for disease control, followed by allo-HCT for curative intent, when feasible.
Disclosures
Patel:Pfizer: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Bristol Myers Squibb: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees. Cerny:Sorrento Therapeutics: Current equity holder in private company; Veru: Current equity holder in private company; TG Therapeutics: Current equity holder in private company; Vaxart: Current equity holder in private company; Miragen Therapeutics: Current equity holder in private company; Gamida Cell: Current equity holder in private company; Mustang Bio: Current equity holder in private company; Novavax: Current equity holder in private company; Ovid Therapeutics: Current equity holder in private company; Jazz Pharmaceuticals: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Amgen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; AlloVir: Membership on an entity's Board of Directors or advisory committees; Actinium Pharmaceuticals: Current equity holder in private company; Bluebird Bio/2Seventy: Current equity holder in private company; aTyr Pharma: Current equity holder in private company; Dynavax Technologies: Current equity holder in private company; Pfizer: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees. Ramanathan:sanofi: Current holder of stock options in a privately-held company; novartis: Current holder of stock options in a privately-held company; J and J: Current holder of stock options in a privately-held company; moderna: Current holder of stock options in a privately-held company; vertex pharma: Current holder of stock options in a privately-held company; pfizer: Current holder of stock options in a privately-held company; gilead: Current holder of stock options in a privately-held company. Gerber:Novartis Pharmaceuticals Corporation: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; US Patent and Trademark Office: Patents & Royalties: US Patent No. 9,012,215; US Patent and Trademark Office: Patents & Royalties: US Patent No. 10,222,376; US Patent and Trademark Office: Patents & Royalties: US Patent No. 11,209,435.
Author notes
 This icon denotes a clinically relevant abstract
This icon denotes a clinically relevant abstract
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal